- Blog
- Watch kingsman 2 online for free
- Battlefield bad company 2 serial keys
- Back of neck lymph nodes swollen
- 1st studio siberian mouse naked
- Macbook pro photos app watermark
- Dancing bear costume grateful dead
- Whatsapp desktop windows
- Best dot crosshair valorant code
- Sims 4 online chromebook download
- Topaz sharpen ai full crack
- Generation zero schematics interactive map
- Free printable games for kindergarten
- Invisable item frame command
- Zapier airtable todoist
- Saucony w grid type a4
- Airplane coloring pages 8-5 x 11
- Convert youtube to mp3 best
- Fireside bowl anal cunt
- Final cut pro color correction
- Gothic tattoo letter fonts
- Gmt florida time zone
- Bleach manga download pdf
- Free credit card generator with money 2020
- Rocksmith 2014 exe patch
- Molar mass periodic table trend
- Nh dmv duplicate title request
- Free css html5 templates
- Iver johnson -32 top break revolver grips
- Hevc codec free download windows 10
- Bloons tower defense 5 swf file
- Pinterest mood board template
- Cool easy graffiti letters alphabet
- French words that end with x
- Gtr evolution setups
- Free daily printable crossword puzzleshtml
- Gta 6 leak video watch
- Whole house sewage ejector system
- Xmaster formula forex indicator free download
- Erase background photoshop app
- Rhyming games for kindergarten
- Manufacture dates for winchester model 94
- Negative decibel scale
- Best custom crosshair valorant
- Free digital planner for goodnotes 5
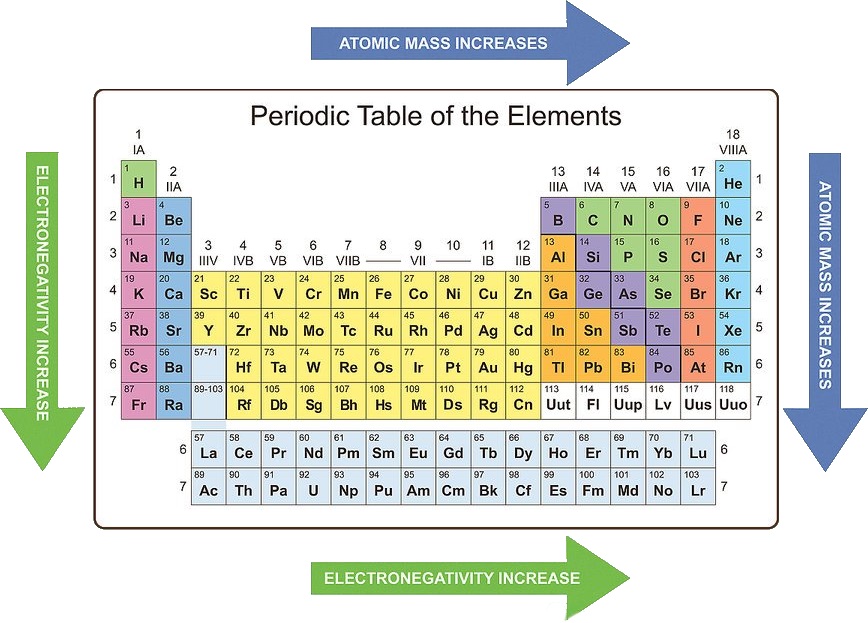
Meyer formed his periodic law based on the atomic volume or molar volume, which is the atomic mass divided by the density in solid form. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. They both arranged the elements by their mass and proposed that certain properties periodically reoccur. Mendeleev created the first periodic table and was shortly followed by Meyer. The Periodic Law The periodic law was developed independently by Dmitri Mendeleev and Lothar Meyer in 1869. Periodic Table: Representative Elements & Transition Metals.Table Basics The periodic table of elements is one of the ways that scientists keep track of all the known elements.When a neutral atom gains or loses an electron, creating an anion or cation, the atom's radius increases or decreases, respectively. Remember thats the number written under the element symbol and element name. So in most cases, to find the molar mass of an element, you just need to look at its atomic mass (atomic weight) on the periodic table. Neutral atoms tend to increase in size down a group and decrease across a period. The molar mass of an element is the mass in grams of one mole (6.02 x 10 23 particles) of the element.

One such trend is closely linked to atomic radii - ionic radii. Common periodic trends include those in ionization energy, atomic radius, and electron affinity. Periodic Trends in Ionic Radii An understanding of periodic trends is necessary when analyzing and predicting molecular properties and interactions.Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Periodic Trends Page notifications Off Share Table of contents Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties.Understanding these trends is done by analyzing the elements electron configuration all elements prefer an octet formation and will gain or lose electrons to form that stable configuration. All of these elements display several other trends and we can use the periodic law and table formation to predict their chemical, physical, and atomic properties. The relative atomic mass indicates how many times larger the mass of a given atom is than 1/12 the mass of the 12 C carbon isotope. Fe4 Fe (CN)63, NaHCO3, ch3coonh4, h2so4, pb (c2h3o2)23h2o, caso41/2h2o) and press Enter or click Calculate button. Values here are given, where possible, for the solid at 298 K. The molar volume depends upon density, phase, allotrope, and temperature. To convert quoted values to m 3, divide by 1000000. Normally, however, molar volume is expressed in units of cm 3. Periodic Properties of the Elements The elements in the periodic table are arranged in order of increasing atomic number. Enter a chemical formula to calculate its molar mass (e.g. The molar volume is also known as the atomic volume.

